Page 26 - MSAM-1-1
P. 26
Materials Science in Additive Manufacturing Spheroidization of 304L SS powder for LPBF process
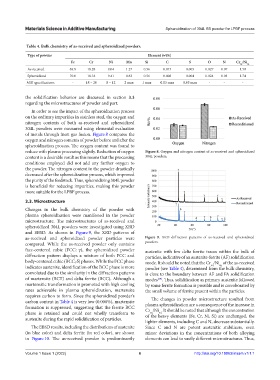
Table 4. Bulk chemistry of as‑received and spheroidized powders.
Type of powder Element (wt%)
Fe Cr Ni Mn Si C S O N Cr /Ni
eq eq
As-received 69.9 18.28 9.04 1.27 0.56 0.015 0.005 0.027 0.07 1.50
Spheroidized 70.0 18.18 9.41 0.83 0.56 0.008 0.004 0.024 0.03 1.74
AISI specifications - 18 – 20 8 – 12 2 max 1 max 0.03 max 0.03 max - - -
the solidification behavior are discussed in section 3.3
regarding the microstructures of powder and part.
In order to see the impact of the spheroidization process
on the ordinary impurities in stainless steel, the oxygen and
nitrogen contents of both as-received and spheroidized
304L powders were measured using elemental evaluation
of metals through inert gas fusion. Figure 8 compares the
oxygen and nitrogen contents of powder before and after the
spheroidization process. The oxygen content was found to
reduce with plasma processing slightly. Reduction of oxygen Figure 8. Oxygen and nitrogen content of as-received and spheroidized
content is a desirable result as this means that the processing 304L powders.
conditions employed did not add any further oxygen to
the powder. The nitrogen content in the powder drastically
decreased after the spheroidization process, which improved
the purity of the feedstock. Thus, spheroidizing 304L powder
is beneficial for reducing impurities, making this powder
more suitable for the LPBF process.
3.3. Microstructure
Changes in the bulk chemistry of the powder with
plasma spheroidization were manifested in the powder
microstructure. The microstructures of as-received and
spheroidized 304L powders were investigated using XRD
and EBSD. As shown in Figure 9, the XRD patterns of
as-received and spheroidized powder particles were Figure 9. XRD diffraction patterns of as-received and spheroidized
compared. While the as-received powder only contains powders.
face-centered cubic (FCC; γ), the spheroidized powder austenite with few delta ferrite traces within the bulk of
diffraction pattern displays a mixture of both FCC and particles, indicative of an austenite-ferrite (AF) solidification
body-centered cubic (BCC; δ) phases. While the FCC phase mode. It should be noted that the Cr /Ni of the as-received
eq
eq
indicates austenite, identification of the BCC phase is more powder (see Table 4), determined from the bulk chemistry,
convoluted due to the similarity in the diffraction patterns is close to the boundary between AF and FA solidification
of martensite (BCT) and delta ferrite (BCC). Although a modes . Thus, solidification as primary austenite followed
[39]
martensitic transformation is promoted with high cooling by some ferrite formation is possible and is corroborated by
rates achievable in plasma spheroidization, martensite the small volume of ferrite present within the particles.
requires carbon to form. Since the spheroidized powder’s
carbon content in Table 4 is very low (0.008%), martensite The changes in powder microstructure resulted from
formation is suppressed, suggesting that the ferrite BCC plasma spheroidization are a consequence of the increase in
Cr /Ni . It should be noted that although the concentration
phase is retained and could not wholly transform to of the heavy elements (Fe, Cr, Ni, Si) are unchanged, the
eq
eq
austenite during the rapid solidification of particles.
lighter elements, including C and N, decrease substantially.
The EBSD results, including the distributions of austenite Since C and N are potent austenitic stabilizers, even
(in blue color) and delta ferrite (in red color), are shown minor deviations in the concentrations of both alloying
in Figure 10. The as-received powder is predominantly elements can lead to vastly different microstructures. Thus,
Volume 1 Issue 1 (2022) 6 http://doi.org/10.18063/msam.v1i1.1