Page 52 - IJB-2-2
P. 52
Rheological study on 3D printability of alginate hydrogel and effect of graphene oxide
layer. Subsequent layers were added layer-by-layer in the gel point: both G'(ω) and G"(ω) are proportional to
the vertical axis. ωn (0 < n < 1) at sufficiently low frequencies, ω. The
definition of the gel point by this power-law is excel-
3. Results and Discussion lent because the gelation variable will lose its depen-
3.1 Sol-gel Transition dency of frequency at the gel point. Several works
have shown that this method is reliable and valid for
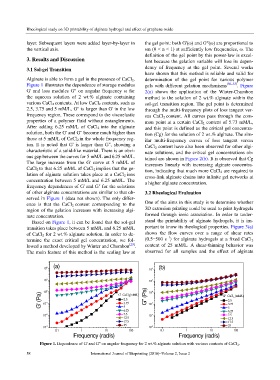
Alginate is able to form a gel in the presence of CaCl 2. determination of the gel point for various polymer
Figure 1 illustrates the dependence of storage modulus gels with different gelation mechanisms [30–33] . Figure
G' and loss modules G" on angular frequency ω for 2(a) shows the application of the Winter-Chambon
the aqueous solution of 2 wt.% alginate containing method to the solution of 2 wt.% alginate within the
various CaCl 2 contents. At low CaCl 2 contents, such as sol-gel transition region. The gel point is determined
2.5, 3.75 and 5 mM/L, G" is larger than G' in the low through the multi-frequency plots of loss tangent ver-
frequency region. These correspond to the viscoelastic sus CaCl 2 content. All curves pass through the com-
properties of a polymer fluid without entanglements. mon point at a certain CaCl 2 content of 5.73 mM/L,
After adding 6.25 mM/L of CaCl 2 into the alginate and this point is defined as the critical gel concentra-
solution, both the G' and G" become much higher than tion (Cg) for the solution of 2 wt.% alginate. The sim-
those at 5 mM/L of CaCl 2 in the whole frequency reg- ilar multi-frequency curves of loss tangent versus
ion. It is noted that G' is larger than G", showing a CaCl 2 content have also been observed for other algi-
characteristic of a solid-like material. There is an obvi- nate solutions, and the critical gel concentrations ob-
ous gap between the curves for 5 mM/L and 6.25 mM/L. tained are shown in Figure 2(b). It is observed that Cg
The large increase from the G' curve at 5 mM/L of increases linearly with increasing alginate concentra-
CaCl 2 to that 6.25 mM/L of CaCl 2 implies that the ge- tion, indicating that much more CaCl 2 are required to
lation of alginate solution takes place at a CaCl 2 ions cross-link alginate chains into infinite gel networks at
concentration between 5 mM/L and 6.25 mM/L. The a higher alginate concentration.
frequency dependences of G' and G" for the solutions
of other alginate concentrations are similar to that ob- 3.2 Rheological Evaluation
served in Figure 1 (data not shown). The only differ-
ence is that the CaCl 2 content corresponding to the One of the aims in this study is to determine whether
region of the gelation increases with increasing algi- 3D extrusion printing could be used to print hydrogels
nate concentration. formed through ionic association. In order to under-
Based on Figure 1, it can be found that the sol-gel stand the printability of alginate hydrogels, it is im-
transition takes place between 5 mM/L and 6.25 mM/L portant to know its rheological properties. Figure 3(a)
of CaCl 2 for 2 wt.% alginate solution. In order to de- shows the flow curves over a range of shear rates
–1
termine the exact critical gel concentration, we fol- (0.5−500 s ) for alginate hydrogels at a fixed CaCl 2
lowed a method developed by Winter and Chambon [29] . content of 25 mM/L. A shear-thinning behavior was
The main feature of this method is the scaling law at observed for all samples and the effect of alginate
2 (a) (b)
10 10 2
10 1 10 1
10 0 G' CaCl (mM/L) 10 0 G'' CaCl (mM/L)
G' (Pa) 10 -1 2.5 2 10 G'' (Pa) -1 2.5 2
3.75
3.75
5
6.25 5
6.25
10 -2 7.5 10 -2 7.5
12.5 12.5
17.5 17.5
25 25
10 -3 10 -3
0.1 1 10 100 0.1 1 10 100
Frequency (rad/s) Frequency (rad/s)
Figure 1. Dependence of G' and G" on angular frequency for 2 wt.% alginate solution with various contents of CaCl 2 .
58 International Journal of Bioprinting (2016)–Volume 2, Issue 2